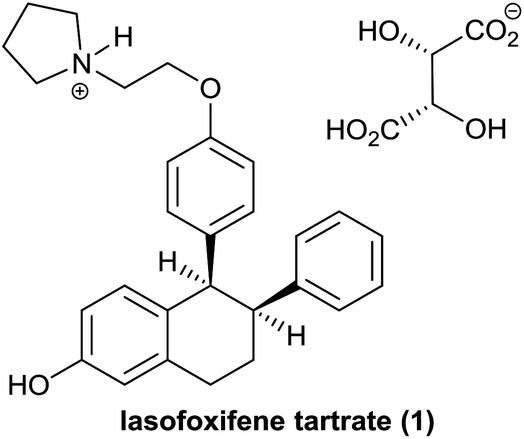
Cyclic Tetrapeptides from Nature and Design: A Review of Synthetic Methodologies, Structure, and Function | Chemical Reviews
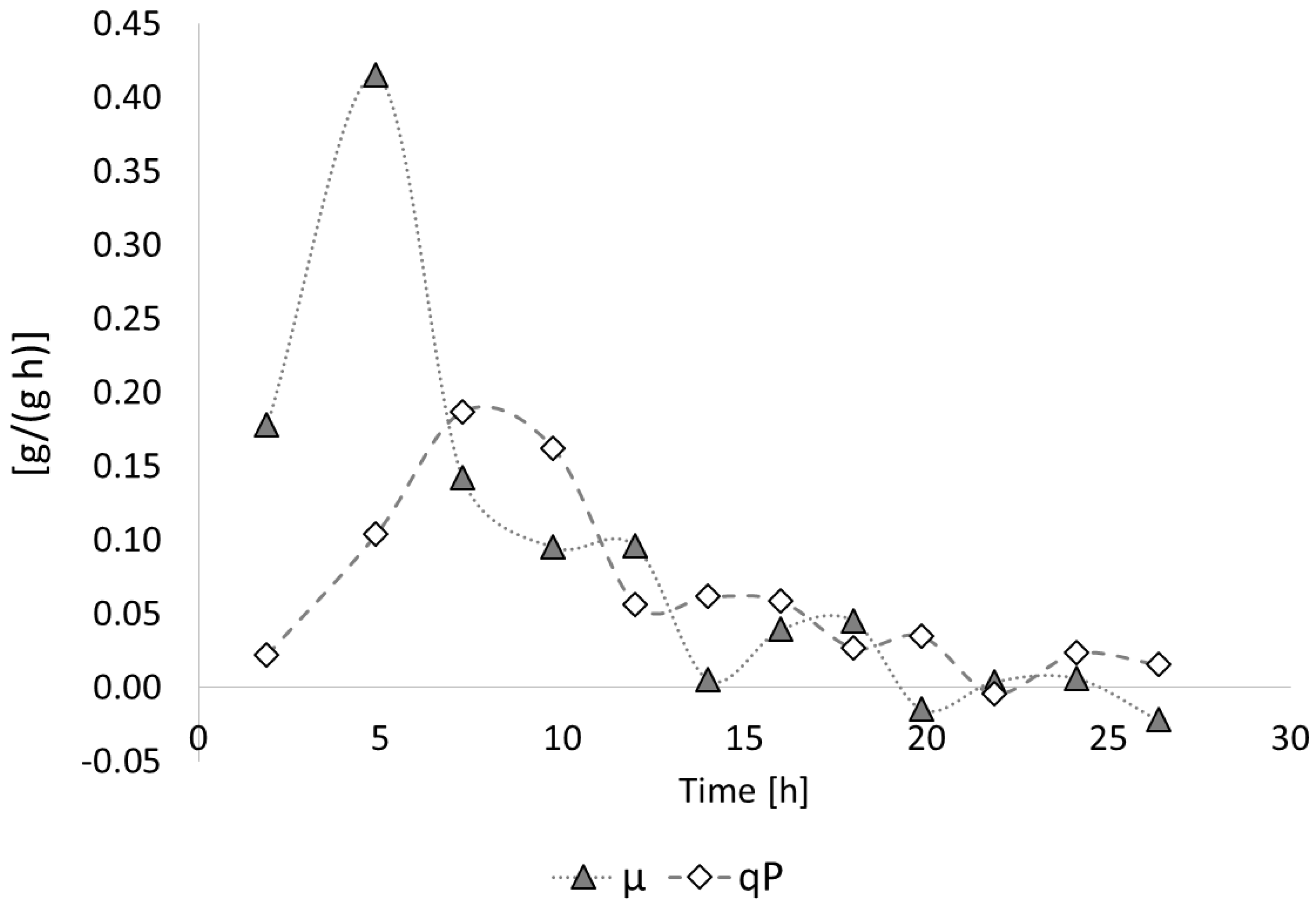
Bioengineering | Free Full-Text | Fed-Batch Synthesis of Poly(3-Hydroxybutyrate) and Poly(3-Hydroxybutyrate-co-4-Hydroxybutyrate) from Sucrose and 4-Hydroxybutyrate Precursors by Burkholderia sacchari Strain DSM 17165

Cyclic Tetrapeptides from Nature and Design: A Review of Synthetic Methodologies, Structure, and Function | Chemical Reviews

Solvent Based Hydrogen Bonding: Impact on Poly(3-hexylthiophene) Nanoscale Morphology and Charge Transport Characteristics | ACS Nano

Antibacterial Natural Products in Medicinal Chemistry—Exodus or Revival? - von Nussbaum - 2006 - Angewandte Chemie International Edition - Wiley Online Library

Bioengineering | Free Full-Text | Fed-Batch Synthesis of Poly(3-Hydroxybutyrate) and Poly(3-Hydroxybutyrate-co-4-Hydroxybutyrate) from Sucrose and 4-Hydroxybutyrate Precursors by Burkholderia sacchari Strain DSM 17165

From Noncovalent Chalcogen–Chalcogen Interactions to Supramolecular Aggregates: Experiments and Calculations | Chemical Reviews
Recent advances in visible light-activated radical coupling reactions triggered by (i) ruthenium, (ii) iridium and (iii) organic photoredox agents - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D1CS00311A